Sunosi (solriamfetol) for narcolepsy
What is Sunosi for narcolepsy?
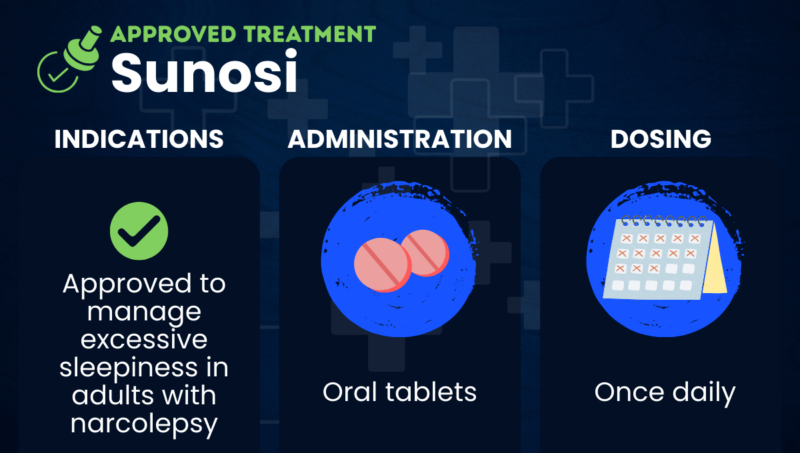
Sunosi (solriamfetol) is an oral medication approved to improve wakefulness in adults with excessive daytime sleepiness associated with narcolepsy.
People with narcolepsy experience uncontrollable urges to sleep throughout the day because their brains can’t properly regulate sleep-wake cycles.
Sunosi works to boost wakefulness by increasing the levels of two brain signaling molecules — dopamine and norepinephrine — that help regulate alertness.
It is marketed by Axsome Therapeutics in the U.S. and Canada, and by Pharmanovia in the European Union (EU). In all of these regions, Sunosi is also indicated for the treatment of daytime sleepiness in people with obstructive sleep apnea. Sunosi’s developers are also evaluating the therapy’s potential use for a range of other conditions.
Therapy snapshot
| Brand name | Sunosi |
| Chemical name | Solriamfetol |
| Usage | Used to improve wakefulness in people with narcolepsy |
| Administration | Oral tablets |
Who with narcolepsy can take Sunosi?
In the U.S., Sunosi is approved to improve wakefulness in adults with excessive daytime sleepiness associated with narcolepsy. It is approved for similar indications in the EU and Canada.
Sunosi is contraindicated, or should not be used, by people currently using a monoamine oxidase inhibitor — a class of antidepressant medications — or who have used one in the last two weeks.
It is also not recommended for people with end-stage kidney disease and should generally be avoided in people with unstable cardiovascular conditions or serious heart issues.
How is Sunosi administered in narcolepsy?
Sunosi comes in the form of oral tablets that should be taken once daily upon awakening. The recommended starting dose is 75 mg, which may be slowly increased to a maximum of 150 mg as needed and tolerated.
Dose adjustments may be needed for people with moderate or severe kidney impairment.
Sunosi should not be taken within nine hours of planned bedtime, as it may interfere with nighttime sleep.
Sunosi in narcolepsy clinical trials
The approval of Sunosi for adults with narcolepsy was largely supported by data from a single Phase 3 clinical trial (NCT02348593). The study involved 239 adults with narcolepsy, who were randomly assigned to receive Sunosi (75, 150, or 300 mg) or a placebo once daily for three months. Study data showed that:
- at the two highest doses, Sunosi was associated with significant reductions in daytime sleepiness, which were evident after a week and sustained throughout the trial
- in an objective test of daytime wakefulness, patients on Sunosi demonstrated significant improvements in the ability to stay awake compared with the placebo
- in a subjective measure, patients on Sunosi reported significantly greater reductions in sleepiness during daily life activities
- a significantly higher proportion of patients who received 150 or 300 mg of Sunosi perceived the change in their symptoms as improved relative to the placebo group
Nighttime sleep was unaffected by Sunosi treatment.
Sunosi side effects
The most common side effects of Sunosi include:
- headache
- nausea
- decreased appetite
- insomnia
- anxiety
The medication’s prescribing information also comes with warnings for potentially serious side effects, including:
- elevations in blood pressure or heart rate
- psychiatric problems, including anxiety, insomnia, and irritability
Heart rate and blood pressure will be monitored at treatment onset and periodically thereafter. High blood pressure must be controlled before treatment starts.
Sunosi should be used with caution in people with existing cardiovascular issues, who are using other medications that can affect blood pressure or heart rate, or who have preexisting psychiatric conditions. The dose of Sunosi may need to be lowered, or the therapy may have to be discontinued entirely if cardiovascular or psychiatric problems develop and can’t be controlled.
Patients should also inform their healthcare team about all medications they are using, as Sunosi could interact with some of them.
Although it is low, Sunosi has potential for abuse. Patients will be monitored for a history of drug abuse or signs of Sunosi misuse/abuse.
Narcolepsy News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by 
